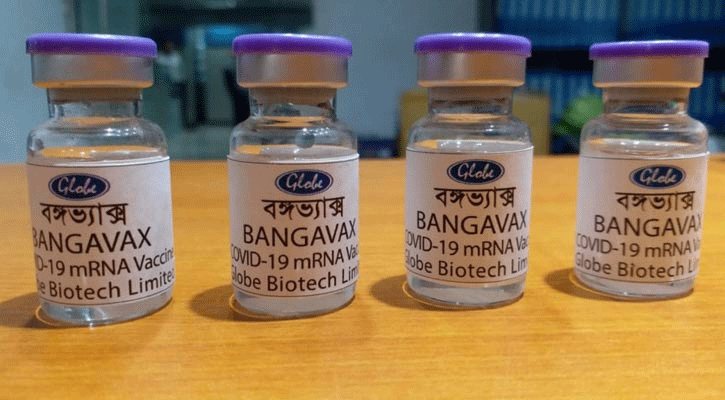
‘Bangavax’ approved for human trial
Staff Correspondent: The Bangladesh Medical Research Council (BMRC) approved in principle for human trial of ‘Bangavax’ corona vaccine developed by Globe Biotech Limited.
“We received ethical permission. Now we will submit the protocol to DGDA (Directorate General for Drug Administration) for clinical trial,” said Dr. Md Mohiuddin, Globe Biotech Senior Manager (Quality and Regulation).
Once approved, the Bangavax vaccine will be administered to humans, he added.
Globe Biotech Limited, the manufacturer of Bangavax, claims that the preliminary results show Bangavax vaccine is 100% effective against 11 variants of corona, including the Delta.
The company further claims that the animal trial of Bangavax vaccine has been successfully completed. The trial report has also been submitted to BMRC.
According to Globe Biotech authorities, Bangavax vaccine is made with natural pure mRNA (Messenger ribonucleic acid). As a result, it has the potential to be the safest and most effective.
Bangavax vaccine is one dose. If approved, the demand for this vaccine will also be created abroad.
Globe Biotech Limited announced the invention of the vaccine for the first time in the country on July 2, 2020. About three and a half months later, on October 15, the World Health Organization (WHO) included three Globe Biotech vaccines in its list of candidates for approval. Globe Biotech is the only company in the world to have the top three vaccine approval lists. The protocol was submitted to BMRC on January 17 this year for the first and second phase ethical test of Bangavax. The revised protocol was then submitted on 16 February as per the demand of BMRC. On June 22, the BMRC allowed for Bangavax human trails subject to trail on monkeys or chimpanzees first. On August 1, the company began trial on monkeys, which ended on October 21.
President Joe Biden tests positive for COVID-19 while campaigning in Las Vegas, has ‘mild symptoms’
International Desk: President Joe Biden tested positive for COVID-19 while traveling Wedne…